|
(More detailed calculations give a value of Z eff = 1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. and Nitrogen (Atomic radii) (b) Potassium and Chlorine (Electronegativity) (c) Magnesium and Calcium (Valency) Give reason for your answer in each case. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately 1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. Fluorine, 42 pm, Chromium, 166 pm, Rubidium, 265 pm.Text lists sorted by: Value Atomic Number Al. Neon, 38 pm, Silver, 165 pm, Barium, 253 pm. of the Group 2 elements - beryllium, magnesium, calcium, strontium and barium. Potassium (K) has a greater atomic radius than that of Calcium (Ca) because when we move down a group, the atomic radius increases and when we move from. Helium, 31 pm, Cadmium, 161 pm, Praseodymium, 247 pm. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from 3 to 10. Trends in atomic radius, first ionisation energy, electronegativity and. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. Hence, calcium has higher atomic radius than magnesium. 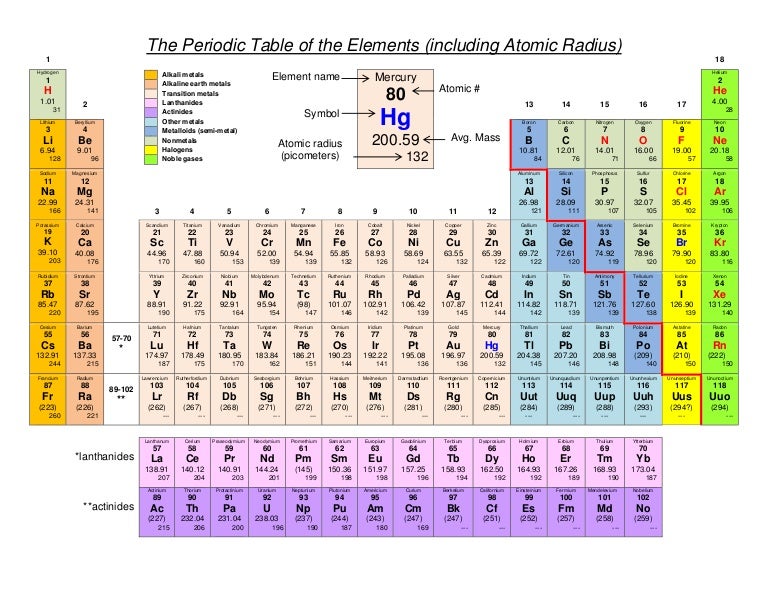
For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. On moving down the group, the atomic radii increase as additional electron shells are added. Depending on the definition, the term may apply only to isolated atoms, or also to atoms in. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. \( \newcommand\)) experienced by electrons in the outermost orbitals of the elements. The atomic radius of a chemical element is the distance from the center of the nucleus to the outermost shell of an electron.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
